About Esco Aster
Esco Aster is a vertically integrated bioinnovation venture accelerator delivering end-to-end “idea-to-exit” contract solutions—from consultancy and research to CMC development, manufacturing, testing, clinical development, business development, strategic capital and enterprise support.
We provide phase-appropriate capabilities that scale with our collaborators, sponsors, and partners – from early development through GLP Tox, clinical, pilot, demonstration and full commercial production.
Founded by scientists, we empower fellow innovators to translate benchwork and intellectual property into real-world impact.
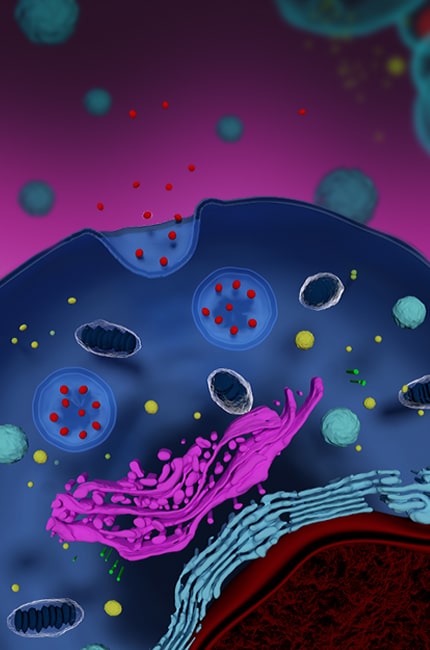
Through our platforms, we serve the broader bio-based deep tech bioeconomy—spanning life-saving diagnostics, therapeutics, vaccines, dermatology, dermocosmetics/cosmeceuticals, medical aesthetics/ injectables/coatings, nutraceuticals, and cellular agriculture — delivering high-yield, cost-effective solutions aligned with self-sufficiency and health technology assessment frameworks.
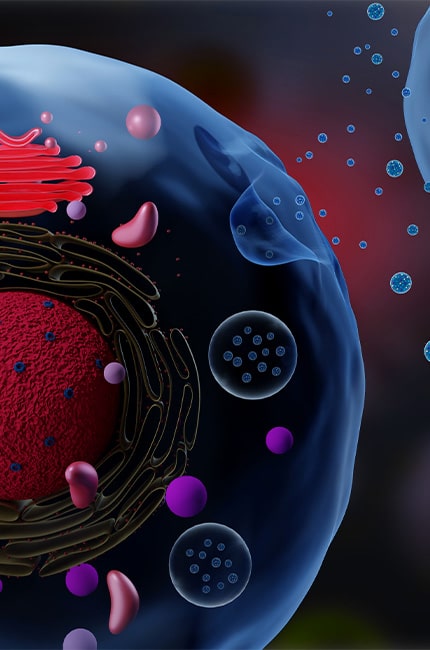
Our patented, best-in-class manufacturing platforms simplify complexity, enabling reliable, linearly scalable outcomes. Our technologies have been successfully deployed in multiple commercial human and animal vaccines, dermocosmetics, as well as in cell-based nutrition and clinical-grade stem cells and exosomes.
We continuously invest in advancing manufacturing science to address unmet clinical needs, unlocking bottlenecks across existing and emerging modalities from concept to cure.
Together with our venture ecosystem, we champion an interconnected One World. One Health approach—bringing innovation to life for people, planet and profit.
Join us in redefining nutrition and medicine for current and future generations.
Esco Aster.
One World Biosolutions for One Health.
About Us
















.webp)