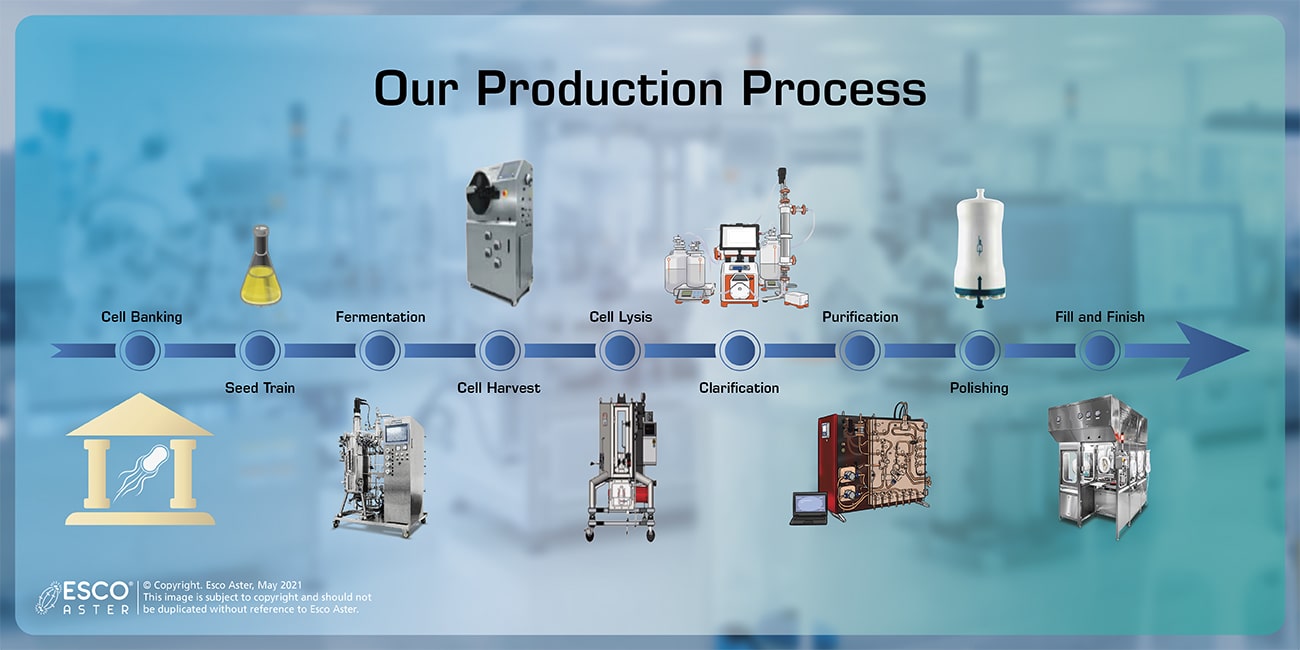
Our Production Process
Esco Aster’s state-of-art facility has an end-to-end, single-use bioprocessing capacity to manufacture plasmid of the highest grade of purity following the GMP guidelines FDA 21 CRF Part 820. This facility includes ISO standards cleanrooms, bioprocessing equipment of cutting edge technology coupled with our experts, and in-house product release testing capabilities. Moreover, the use of single-use technology eliminates the possibility of cross-contamination between products and batches. We maintain highest standard of cleanliness of the facility by following EU GMP annex 1 guideline for environmental monitoring.
Esco Aster has a facility to perform microbial fermentation, cell harvesting by continuous centrifugation, cell lysis by alkalization, clarification in tangential flow filtration, purification and polishing in liquid chromatography. All the equipment and core technologies are selected and validated for single-use operation. Esco Aster has branded isolator system for bulk filling of the final product into sterile packaging.