Overview
Extracellular vesicles or EVs have emerged as a new class of nanovehicles, comprising of nanovesicles such as exosomes (30 – 200 nm), microvesicles (MVs) (50 nm – 1um), and apoptic bodies (500 – 1000 nm). EVs have a remarkable potential as novel candidates for their efficiency as a vehicle to deliver therapeutics/drugs as cargo to a disease site and are key for intercellular communication.
EV-based therapies are valuable for their role in immune function, treating infectious diseases and cancer, as well as neurological disorders. Given that cells produce and secrete EVs naturally, in cases of EV-secreted stem cells, its health benefit for tissue regeneration triggers a significant interest in the medical field. Their stable size and composition enable them to protect their ‘cargo’ against degradation and can even cross biological barriers, like the blood-brain barrier, that makes them superior to other nanocarriers.
And while the presence of challenges remains, what differentiates the medical or therapeutic characterization of EVs is likely its protein and mRNA characterization. Different methods to characterize EVs are a work in progress, including the novel direct stochastic optical reconstruction microscopy. Esco Aster makes clinical translation of EVs possible, for treating fatal diseases, as companies explore and develop approaches to upscale into manufacturing.
Click here to learn more about our EV manufacturing and product offerings.| Upstream Processing |
|---|
| Full Capable and automated technologies for exosome generation and expansion. |
| Origin |
| Cell line selection and isolation. |
| Expansion and Engineering |
| Large-scale 3D Tide Motion systems (adherent cell culture) |
| Viral vector transfection methods |
| EV loading through different methods |
| Downstream Processing |
|---|
| Isolation and loading technologies for exosome product. |
| Tangential flow filtration (TFF) or tangential flow depth filtration (TFDF) for initial clarification |
| Continuous Chromatography for purification |
| Formulation |
| Compounding of Product for target excipient |
| Additional parameters for final exosome composition |
| Delivery |
| Shipping and storage |
| Formulation and Filling |
|---|
| Final path for exosome manufacturing |
| Formulation Development |
| Buffer and Excipient selection for optimal formulation |
| Lyophilization of exosome thru AsterFreeze™ for storage |
| cGMP Manufacturing and QC Testing |
| Manufacturing in cGMP facility |
| QC studies and implementation of USP methods |
| QC release and stability |
| Storage thru AsterVault™ |
Continuous Exosome/Extracellular Vesicles/ Engineering
Continuously Engineered Exosomes/Extracellular Vesicles (CEEVs) as precision medicine satisfies the needs of well-funded pharmaceutical and biotechnology companies.
Esco Aster aims to actively partner with major cell and gene therapy businesses in the rapidly emerging field of EVs.
Esco Aster leverages our platform adherent manufacturing capability and know-how to develop Regenerative medical grade naïve extracellular vesicles (MNEVs) and Continuously Engineered Exosomes/Extracellular Vesicles CEEVs (CEEVs).
Regenerative medical grade naïve extracellular vesicles (MNEVs) are not loaded with any cargo can be used to treat conditions that affect health across an entire life span, including orthopedics, cartilage repair, rare conditions such as avascular necrosis, sensory acuity, and skin conditions. These are normally manufactured and filed as medical devices and cosmeceuticals. Additionally, quantitative data on the efficacy is provided with our in-house in- vivo and in-vitro skin testing methods.
Continuously Engineered Exosomes/Extracellular Vesicles CEEVs (CEEVs)are designed as precision diagnostics, delivering active ingredients into specific cells with precision targeting as Cell- gene therapies for broad diseases ranging from single base pair genetic disorders, orthopedics, neuro and cartilage degeneration, virus infection, cancer, metabolic syndromes. With our CEEV technologies, we are vertically integrated with cGMP manufacturing of both, CEEVs and payloads for precision targeting.
This is enabled by the following three core technologies:
-
Precision Targeting
Single Domain Antibodies (Camelid Naïve Library / Sharkelids) are conjugated to the surface of the EV for precision targeting into specific cells to unload cargo and taken up into cells.
-
Cargo loading
Active components, termed cargoes such as small molecules, RNA, DNA, macromolecules, peptides or proteins are loaded into EVs for delivery. These cargoes are synthesized by flow chemistry, oligonucleotide synthesis, fermentation, or other means. Loaded cargo is then purified exploiting techniques like filtration and chromatography, with sound analytics in place to ensure identification of loaded and unloaded EVs before formulation & drug product filling in vials / bags.
-
Continuous Adherent Tide Motion manufacturing
Esco Aster utilizes our adherent Tide Motion platform for continuous upstream to downstream bioprocessing, chaining continuous harvesting to continuous downstream processing for process intensification. This enables us to provide EVs at higher yields and more affordable cost compared to even suspension culture.
![]()
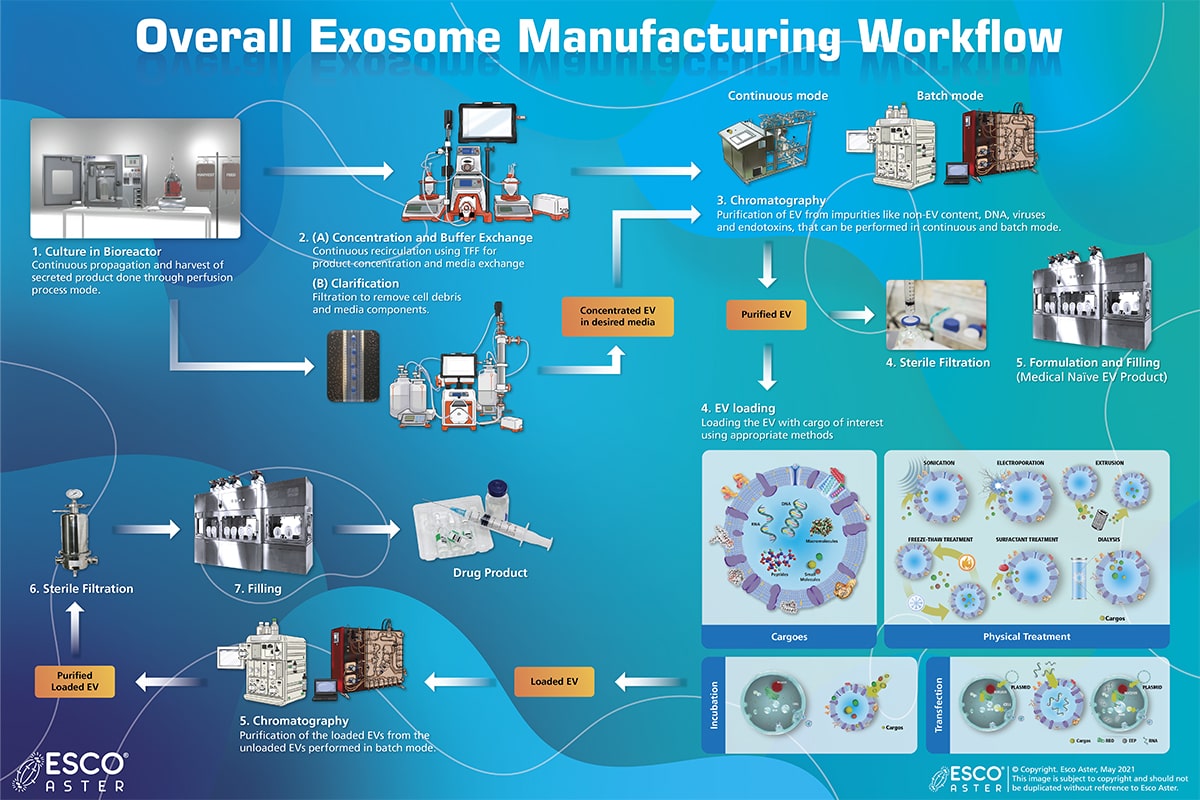
Tide Motion bioreactor is used for the continuous production of exosomes or EVs. This system is equipped with different process modes for ease of feed of fresh culture medium and harvest of secreted product. The harvested medium is clarified via filtration to remove cell debris and residual media components, post which, it is concentrated and buffer exchanged to desirable media via tangential flow filtration (TFF) (. Non-EVs, DNA, viruses, and endotoxins are then removed via chromatographic purification using monoliths, performed at high binding capacities and flow rates to ensure minimum process time with maximum efficiency. Purified EVs is formulated into a medical naïve EV product or be loaded with a specific cargo.
The cargoes can be of various types including RNA, DNA, small molecules, peptides macromolecules etc.
Loading the EV with cargo can be done via various techniques, involving (1) incubation with the cell which releases the target exosomes, (2) transfection via plasmids or directly into the cells, (3) Physical treatments such as electroporation, sonication, freezing, surfactant treatment, dialysis, extrusion etc. The loaded EVs is separated from the unloaded EVs via chromatography, followed by sterile filtration and formulated to the final product.
To discuss our capabilities in EV manufacturing, contact us at [email protected]