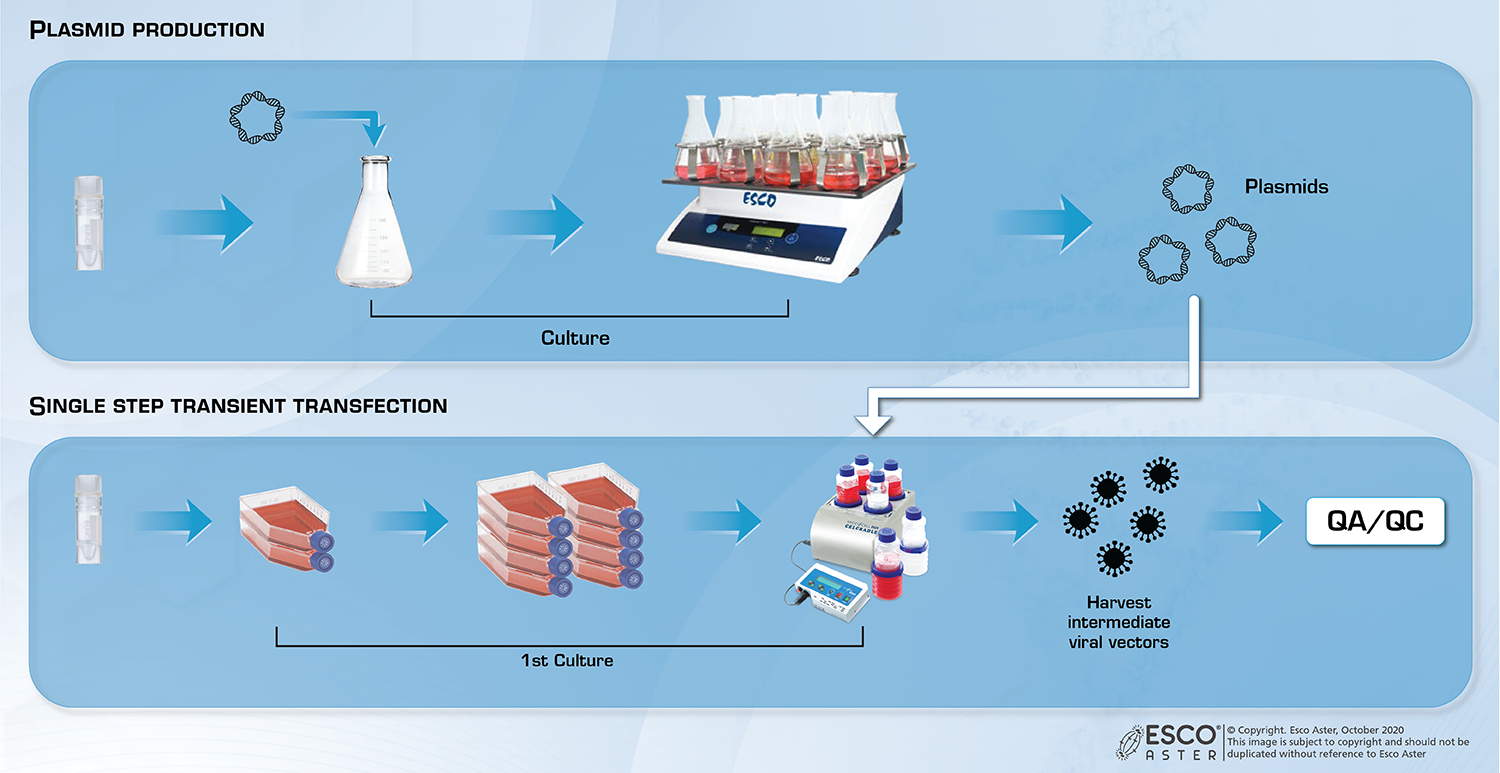
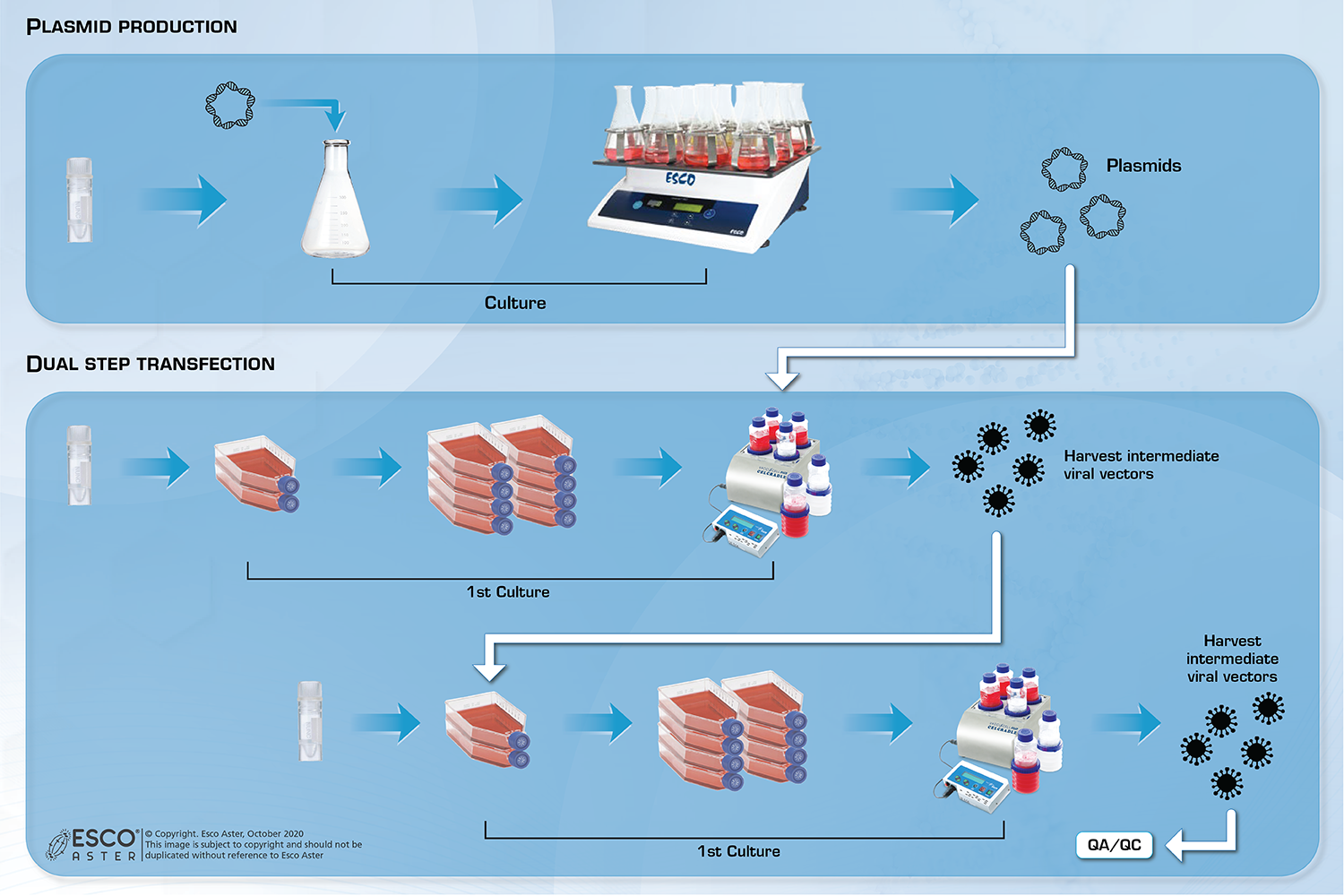
Role of Viral Vectors:
Viral vectors are commonly used in the field of gene therapy to deliver desired gene into the cells. The use of viral vector was adopted due to its efficiency in infecting cells. Due to the increasing number of leads in the field, there bound to have a high demand for these viral vectors for varies clinical trials. Thus, there will be a bottleneck in the viral vector production.
Viral Vector Production Services
Efficient, large-scale production of lentivirus, retrovirus, adenovirus-associated, adenovirus stocks compared to cell factories and other 2D culture systems using producer cell lines such as:
- HEK-293T
- Phoenix-GP
- PG13

Key Application:
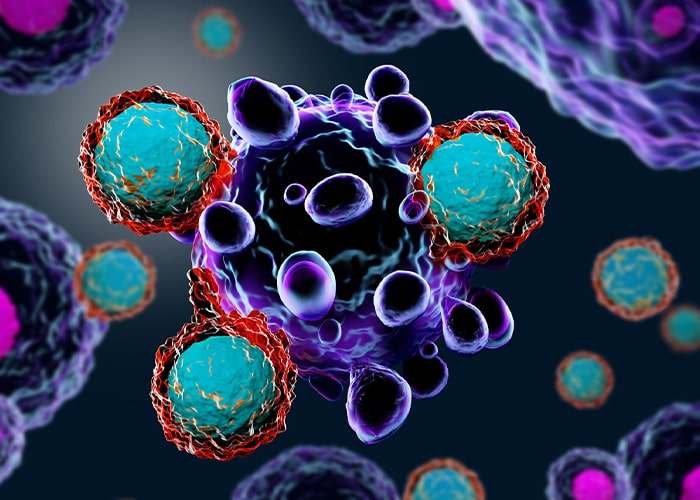
CAR-T Cell Therapy /
CAR-Immune Cell Therapy
CAR-T cell therapy / CAR-Immune Cell Therapy is an immunotherapy to treat cancer. T cells/ Immune cells are re-engineered with the help of viral vector to expression surface receptors that can specifically recognize and target cancer cells.