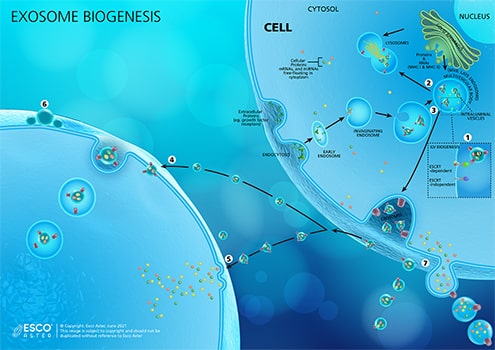
Figure 1. Exosome Biogenesis. (1) Invagination of the endosomal membrane generates Intraluminal Vesicles or ILVs in MVBs, which contain proteins, mRNAs, and miRNAs from the cytoplasm. This process can be done by either ESCRT-dependent (ESCRT stands for endosomal sorting complexes required for transport) or ESCRT-independent mechanisms. ILVs accumulated within the lumen of MVBs have three distinct fates: delivering content for the biogenesis of lysosome-related organelles; (2) fuse with lysosome, which degrade multivesicular endosomes (MVE) contents; or (3) fuse with plasma membrane where ILVs liberated into the extracellular space are released as exosomes. For exosomes to reach their destination and elicit a response from recipient cells, they may (4) be taken up by the target cell’s endocytic pathway, delivering the exosome via back-fusion event, or (5) fusing into the cell’s plasma membrane and releasing its contents directly to the cytosol. (6) Alternatively, exosomes may attach to the surface of the target cell to elicit a signaling response. (7) Membrane-derived vesicles such as ectosomes and microvesicles among others, are also secreted by the cell.
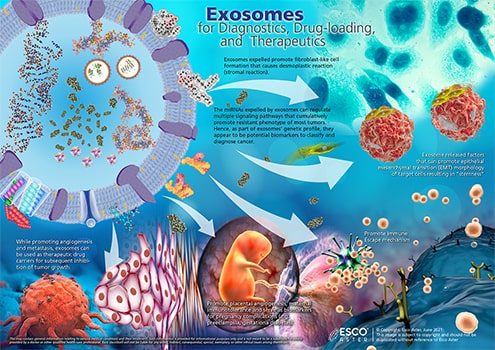
The emerging role of exosomes as a natural nano-vehicle, which were once thought of as cell’s disposal system, offers a big potential in therapy (Figure 2). They are virtually in every cell type of the body ranging from 30-120 nanometers in size. For example, exosomes released by stem cells may deliver a cargo that inhibits or promotes tumor growth, aid in healing and regenerations, and more. Those that are produced by immune cells are known to have regenerative and protective effects in conditions such as cardiovascular disease and stroke. This only means that different cell types secrete exosomes with unique molecular delivery that performs, specifically in a targeted area. This unique property is a key consideration for manufacturing exosomes for therapeutic use.
Figure 2. Exosomes for Diagnostics, Drug-loading, and Therapeutics. EVs, especially exosomes, have been utilized as an ideal drug delivery vehicle for restoring immune function, treating infectious diseases, and even in cancer.
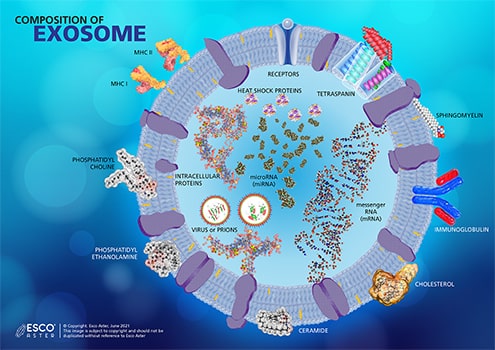
Every exosome has a unique content derived from its parent cell. They contain the following but are not limited to various types of ribonucleic acid (RNA), tetraspanin, different lipids such as cholesterol, sphingolipids, and ceramides, heat shock proteins, integrins, and more (Figure 3). Studies have shown that exosomes secreted by platelets carries prostaglandin which is usually involved inflammatory responses. In general, exosomes facilitate cell-to-cell communication and deliver molecules in a target-specific manner.
Figure 3. Combined Composition of Exosome. The composition of an exosome varies from the cell of its origin. Some cell secretes exosomes within the bi-layered membrane, while others are comprised of different lipids and saturated fatty acid chains.
EVs have powerful therapeutic effects from both naïve and loaded formats with varying payloads and influence cell behaviors. When delivered to the site itself such as in medical conditions of osteoarthritis, they can potentially be used as complementary and alternative medical therapy. EVs aid to alleviate pain or wound healing to accelerate healing. When delivered especially with a targeting platform such as single domain antibodies in intravenous or intramuscular, intrathecal etc., these can home in to target distal tissues or even cross the blood brain barrier which other nanovehicles cannot.
As EVs are naturally occurring they are intrinsically biocompatible and safe for injection into human body. The function of loaded active ingredients and payloads very likely is biomimetic to how EVs function naturally in vivo; hence, holds a vast promise for different modes of therapies (Figure 4). These could range from potentially targeting the tumor micro-environment, palliative effects, entering the lungs, central nervous system, spine, and other areas.
EV loading is typically carried out using various techniques like sonication, which includes electroporation, co-incubation, sonification, water-bath sonification, probe sonification, permeabilization with saponin etc.
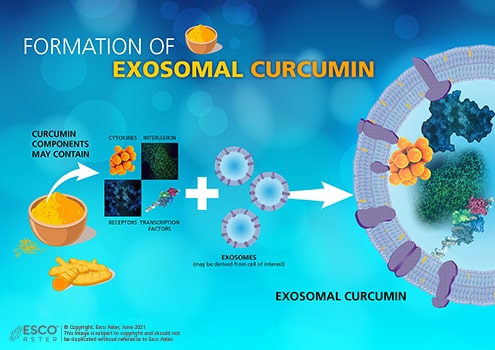
Beyond synthetic payloads, different bioactive payloads from natural compound libraries can be loaded such as curcumin which is the bioactive pigment of turmeric which has polyphenolic-hydrophobic components and can been used for the treatment of a variety of diseases.
Figure 4. Curcumin contains many components that can be extracted for treating disease. Depending on the target, the components will be incorporated into an exosome derived from a chosen cell line. These exosomes will then be released in the extracellular medium after culture and isolated through centrifugation process.
EVs are being widely studied and as of date there are more than 900 clinical trials in varying stages. As these natural vesicles are associated for treating certain types of disease and its potential in clinical diagnosis, current and future research on EVs brings promise to prevention and therapy.
Esco Aster is a scale up and commercialization partner especially for adherent/semi-adherent culture and supplemented by suspension/fermentation for scaling up for EVs/Exosomes.
Capabilities:


Our Partnering Strategy
Esco Aster is actively seeking partnerships with biopharmaceutical companies with interest in bringing exosome-based medicines to their therapeutic areas. We aim to provide faster time to clinic and market to translate EV potential in your target therapeutic area (such as neurology, cancer, or autosomal diseases) into legitimate development plans. Linearly scalable cGMP manufacturing platforms are available to kick-start non-clinical development, GLP Studies, Phase 1, and clinical trials.
What Esco Aster Brings to partnership:
1. Research Services
From biospecimens, spent media and biofluids such as plasma, tissue culture media, cerebrospinal fluid. Esco Aster will isolate and assess quality as per ISEV or client preferred guidelines utilizing spectrophotometry or equivalent techniques. RNA can be further extracted from exosomes and miRNA isolation and sequencing provided. Surface markers can be further analyzed on protein structure and proteomics analysis will provide detailed map to characterize the peptides formed and samples differentially analyzed to determine in silico changes in peptide profile.
Complete exosome profile is then provided as final report.
Esco Aster adapts a bench scale downstream protocols for purification from ultracentrifugation, density gradient centrifugation, and testing of different resins. This also includes different chromatographic steps from size exclusion to others and optimization processes to identify right resins and protocols in achieving purity and recovery whilst still maintaining identity.
2. Clinical-commercial scale manufacturing
Esco Aster have available bioreactor units and platform tools to use and to provide a full CDMO service for EV clinical to commercial scale manufacturing.
This includes:
- Isolation from biofluids or conditioned culture media including platelets, mesenchymal stromal cells, iPSCs, hESCs, other cells (e.g., CAR-T cells) and biogenesis including specific cell culture media to boost expression of EVs.
- Maximum yield from 0.1 L bench-scale to multi-liter bioreactor scale volumes with full preservation of exosome integrity.
- cGMP purification of exosomes for GLP, clinical, medical, cosmeceutical applications
- Media Development including spent media analysis and formulation into serum free media.
3. Specialized payloads and cargo
Esco Aster performs insertion of specially designed nucleic acids to the payloads of EVs: from mRNA, RNAi, and siRNA, which are commonly used to modify genetics of cells. Through the oligosynthesizer, payloads are produced as part of our vertically integrated CDMO service.
4. Precision Targeted delivery
Esco Aster proprietary technology enables the attachment of custom single domain proteins to the surface of EVs. This allows precision targeting of EVs to selected cell types.
5. Analytics & characterization
Esco Aster Analytical Development team provides a comprehensive suite of EV characterization and analysis technologies along with deep experience in encapsulation, formulation, assays, and release.
These focus on physical, biophysical, biochemical, biological assays and include techniques such as single cell flow cytometry. We are evaluating and collaborating on novel technologies for EV Characterization such as dstorm-microscopy as well as other analytical techniques for functional and potency assays.
Esco Aster is open to research collaborations as well to work on novel analytics contact us at [email protected] for collaborations.
Vertically Integrated Manufacturing Technologies
Esco Aster utilizes our adherent Tide Motion platform for continuous upstream to downstream bioprocessing chaining continuous harvesting to continuous downstream processing for process intensification. This enables us to provide EVs at higher yields and more affordable cost than compared to even suspension culture.
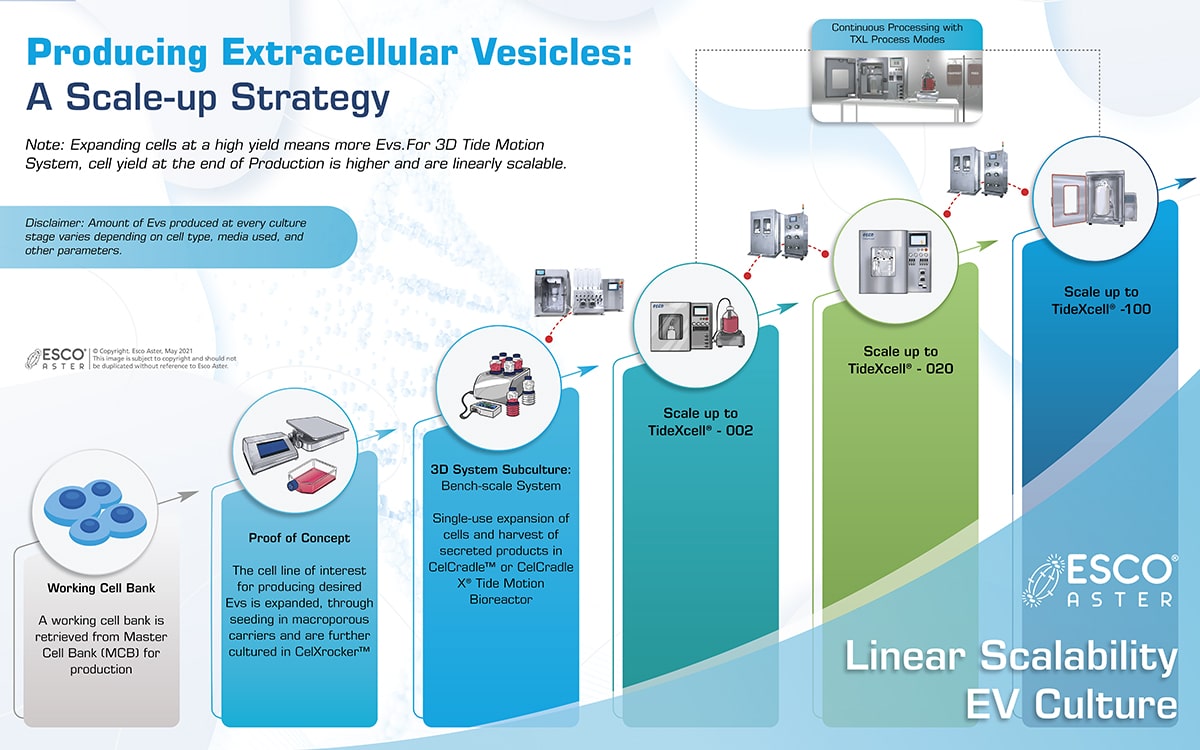
Unlike culturing in microcarriers in suspension, which may suffer from aggregation, non-uniform cell seeding, large clumps, necrotic cores, sludge at bottom of stir tank which tends to be less homogenous in most suspension bioreactors, Tide Motion does not have such limitations. Most multi-layered cell culture-ware do not have its own automated harvesting system, sensors for inline monitoring, not linearly scalable, and will need to scale out. These components are common for most cGMP Quality by Design Manufacturing.
These 2D systems are not linearly scalable and requires scaling out instead of scaling up. While retention of skilled cell culture specialist for 2D culture can be costly, fragile culture ware also tends to result in batch losses resulting in downtime due to clean up and economic losses, especially when they are scaled out up in large batches.
References
- Colao, I. et. al. (2018). Manufacturing exosomes: a promising therapeutic platform. https://doi.org/10.1016/j.molmed.2018.01.006.
- Bianco, N.R. et al. (2009) Therapeutic effect of exosomes from indoleamine 2,3-dioxygenase-positive dendritic cells in collagen-induced arthritis and delayed-type hypersensitivity disease models. https://doi.org/10.1002/art.24229
- Li, V. (2021). Exosomes: a case study in manufacturing a new modality at scale. Biocentury inc.