Research and Development
Esco Aster is currently seeking to discover novel targets expressed on cancer cells and develop high-affinity chimeric antigen receptors using rapid screening assays which in turn capable or attaining high throughput and reproducibility of results.
CAR Construct and Optimization
Empowered by leading technology and years of experience in bioengineering, Esco Aster has executed every strategy and approach to serve a comprehensive CAR design and optimization for our clients.
Since the successful manufacture of CAR-Immune cells is dependent on immune cell receptor gene transfer, genetic modification of these immune cells is one of the most vital steps in generating superior CAR immune cells. To genetically modify these cells, our skillful scientists adopts various methodologies to transfect CAR construction into the desired immune cells.
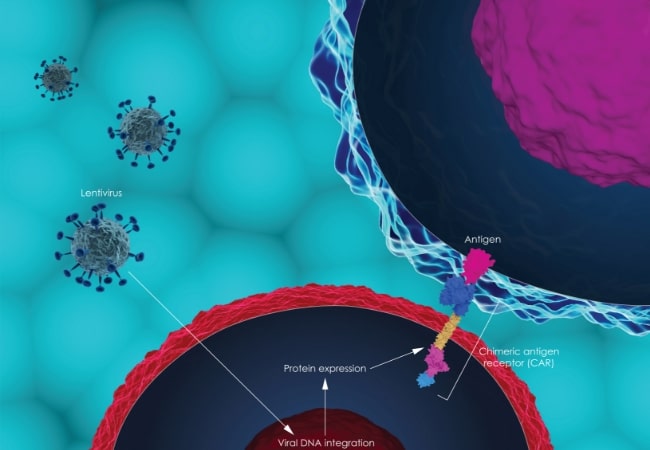
CAR Construct Screening
Screening and evaluating CAR expression is an important step in the production of CAR-T cells. Esco Aster is capable of carrying out specific assays to obtain a multidimensional data set describing CAR and target through kinetic, phenotypic, and functional characterization.
One of which is flow cytometry, utilizing protein L, anti-Fab antibodies, or fluorescent-labeled target antigens as detection methods.
Plasmid Design for Viral Vectors
While plasmids are typically used to encode, propagate, and manipulate genetic information, viruses are a powerful tool for the delivery of this genetic information to cells. Adapting plasmid DNA for use with virus- mediated delivery has many advantages for research. Viruses facilitate the delivery of genetic information to hard-to-transfect cell lines.
Naturally occurring viruses harbor genetic material termed as “viral genomes’, these viral genomes have been adapted into a plasmid-based technology, such that plasmids can be used to create viruses with specific genomes. In other words, instead of a virus infecting a host and giving rise to more virus, at what happens in nature, researchers and scientists can introduce plasmids to a host cell to generate virus. Moreover, these plasmids can be modified to give rise to viral genomes of choice.
Esco Aster’s viral vector manufacturing including lentivirus, retrovirus, adeno-associated virus and many more is well-established using its proprietary Tide Motion technology.
Plasmid Design for Non-Viral Vectors
With the goal of bringing down the prices of CAR-Immune Cell Therapy to make this promising treatment more accessible to a lot of patients, Esco Aster recognizes that viral manufacturing is indeed costly that’s why we are continuously addressing this concern using non-viral vector gene delivery system.
CAR-T cell therapy products produced by non-viral vectors is simpler in the production process and easier to perform quality control comparing with that of viral vectors. At the same time, in terms of storage, the plasmid DNA or mRNA used by the non-viral vector has higher stability, lower storage requirements and longer validity period than the RNA virus of the viral vector system. The advantage of simplified production and lower cost is prominent since CAR-T cells prepared by viral vectors are expensive involving numerous production and quality controls.
Gene Editing
As part of optimizing CAR-immune Cell Therapy for future clinical trials, Esco Aster has been working into addressing the bottlenecks in the manufacturing of autologous products such as T-cell exhaustion, lack of CAR T-cell persistence, and cytokine-related toxicities.
With the ease and accessibility of CRISPR-Cas9-based gene editing, it is possible to address many of these limitations. Accordingly, current research efforts focus on precision engineering of CAR-immune cells with conventional CRISPR-Cas9 systems or novel editors that can install desired genetic changes with or without introduction of a double-stranded break (DSB) into the genome.
CRISPR/Cas9 is a straightforward and effective genome editing tool. By enabling any site-specific genome editing without introducing foreign DNA, Esco Aster can revolutionize your individual project with the help of our well-established platforms and skilled experts.
GLP Grade R&D Service
With Esco Aster's team of experts, we develop custom solutions and offer comprehensive, fully integrated CRDMO services to support its clients' CAR-immune cell therapy projects from the planning stage to commercial manufacturing in strict accordance with the strictest quality, safety, and environmental regulations. Our commitment is to give best-in-class services.

Process Development
Esco Aster is currently seeking to discover novel targets expressed on cancer cells and develop high-affinity chimeric antigen receptors using rapid screening assays which in turn capable or attaining high throughput and reproducibility of results.
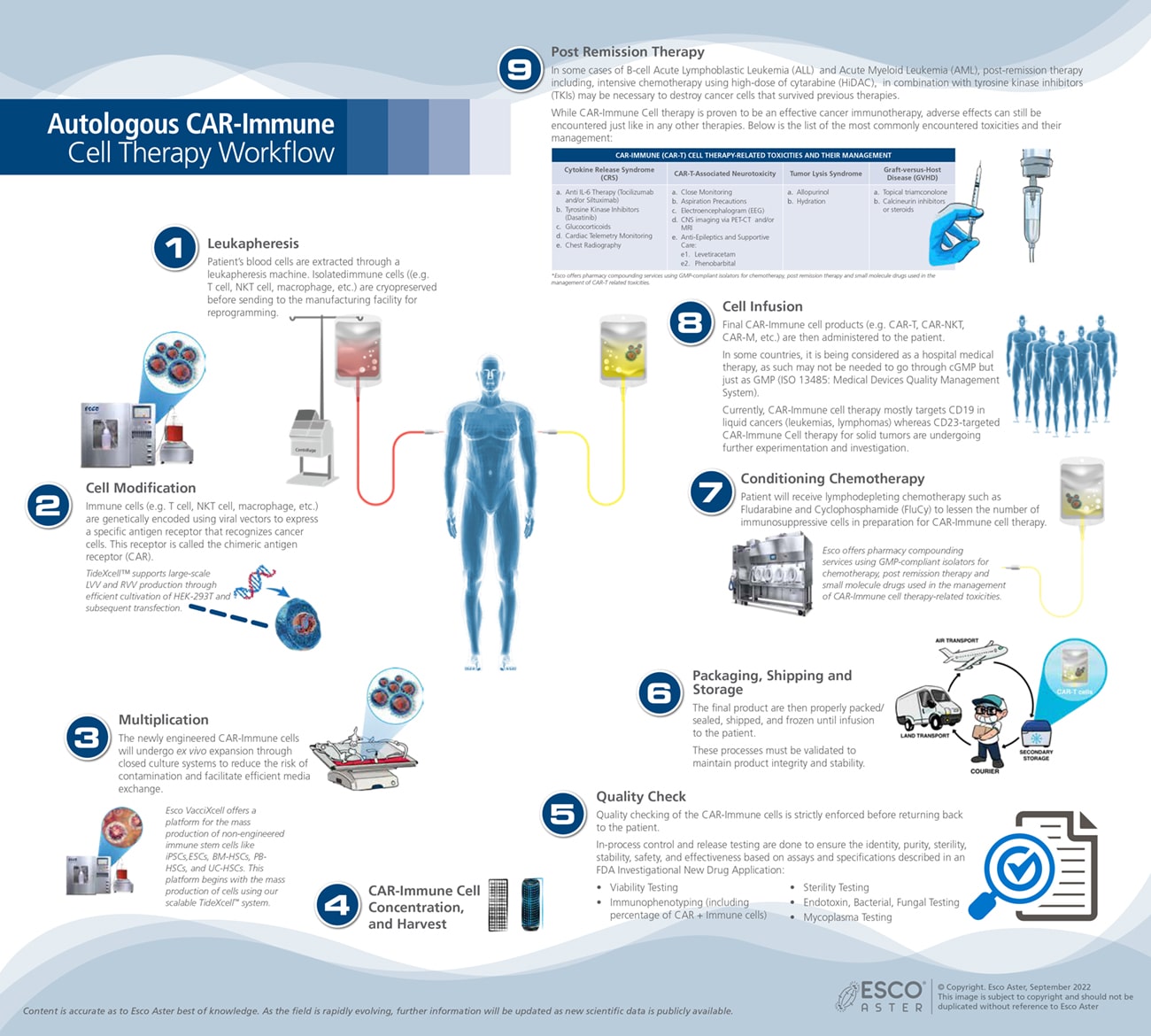
Esco Aster’s process of CAR cell engineering and therapy starts with a closed-system cell isolation procedure. A blood sample is taken from a healthy donor/an autologous patient and white blood cells are separated from other non-leukocyte blood components, through a process called leukapheresis.
Mononuclear cells can then be purified using density gradient centrifugation and analyzed for cell viability and expression of immune effector-cell markers prior to activation and expansion for downstream cell engineering.
Prior to genetic modifications, isolated immune cells require activation and expansion. Esco Aster utilizes versatile and stringent cell activation protocols which is vital as over activation may cause activation-induced cell death.
Esco Aster’s versatile cell transduction/electroporation techniques involve the use of either viral vector on non viral vector technologies based on client’s custom-specific needs.
Our robust lentiviral vector, retroviral vector, adeno-associated viral vector platforms as well as CRISPR/Cas9 technology (non-viral) guarantee the highest success rate of each client’s project.
In order to obtain sufficient amount of CAR Immune cells, a proper duration for T cell expansion ex vivo is necessary. Esco Aster’s rigid expansion protocols have reduced duration of ex vivo culture resulting to lesser cell differentiation and enhanced CAR immune cell therapy efficacy.
Using our proprietary Tide motion bioreactors we are fully-able to produce large-scale lentivirus, retrovirus, adeno-associated virus. Producer cell lines used like HEK293T adapts very well with our bioreactors, reaching a higher cell density for sustained cell growth to allow high titer vector stocks harvested.
With state-of-the-art facility, Esco Aster employs automated cell harvesting and formulation using the latest technology to ensure that 95% cell recovery is attained.
Along the complex supply chain of CAR-immune cell therapy, the cells must remain viable and functional. Ineffective methods of preservation may negatively impact the outcome of the therapy. Esco Aster has developed effective methods of cryopreservation which permit coordination of the therapy with patient care and completion of safety and quality control testing.
Pilot Production Scale
Esco Aster offers high-quality custom manufacturing services covering the entire CAR-T from cell isolation to your end product.
Our commercial production scale culture platform include but not limited to:
- Autologous cells of up to 3L culture with more than 500 million CAR+ cells
- Allogeneic cells of up to 10L culture with more than 1 billion CAR+ cells

Quality Controls
Proper quality control of CAR-immune cells is essential for treatment effectiveness and reproducibility. Esco Aster complies with all CAR-immune cell manufacturing quality controls which cover inspecting the materials used in the production for pathogen contamination, purity, and biological activity (cells, vectors, reagents), and process controls.
We ensure batch-to-batch consistency of the final product by testing the quality of cells during the preparation process. Before product release, our experts make sure that the final product/s meets the specified release criteria which include the following tests/assays:
- CAR+ % (or TCR etc.)
- Cell Viability
- Cell Purity (T cell, NK cells etc.)
- IFN-γ Release
- Sterility
- Mycoplasma Detection
- Vector Copy Number (VCN) Analysis
- Other Phenotype Test per request
- Other Functional Test per request
- Other Impurities Test per request