![mission and vision]()
Transforming Innovation in the Bioeconomy to Advance One World, One Planetary Health
We are dedicated to working with the venture ecosystem to launch, build, and accelerate early-stage companies emerging from leading academic labs, academic medical centres, hospitals, and research institutions focused on cells and their derivatives. We collaborate with fellow venture builders, venture studios, incubators, shared co-working wet labs, knowledge partners, and capital providers.
Collectively, we enable faster fundraising by shortening the time to pilot and clinical-stage products, thereby meeting investment milestones for faster exit and/or commercialization. This enables you to bring your intellectual property to life, supported by Esco Aster’s knowledge-intensive, capex-heavy, and linearly scalable biomanufacturing platforms.
Biotech and startups can therefore remain lean by being knowledge-intensive and capex-light, ensuring maximum financial returns to stakeholders while de-risking through reliance on Esco Aster’s proven first-in-class and first-in-market track record.
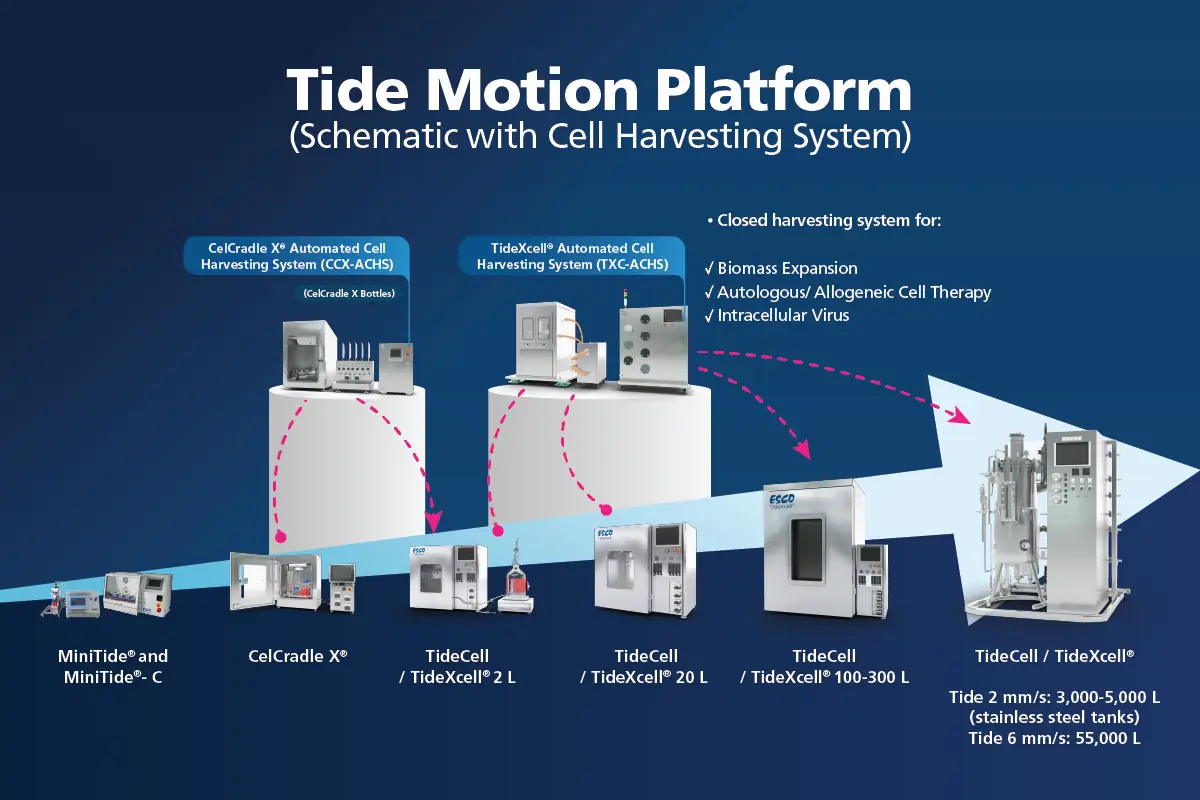
Esco Aster is first in class in cell-based foods (cultivated meat), engineered cancer exosomes targeting HLA-G, and single-use adherent packed-bed bioreactors with automated closed cell harvesting; and first in market for induced Retinal Pigment Epithelium (RPE) from pluripotent stem cells for Dry AMD (Age-related Macular Degeneration).
We focus on vaccines, therapeutic products, cell- and gene therapies, functional nutrition, and technologies at the molecular level that can repair, replace, or regenerate cells, microtissues, tissues, organoids, organs, and/or genes via genetic editing techniques, with the ultimate goal of treating the underlying root causes of disease rather than managing symptoms alone.