From Roller Bottles
to Tide Motion® Bioreactors
Many commercial biologics utilize cells such as adherent CHO for production of Eythropoietin (EPO), Ipilimumab, Interferon beta-1a and others; human and animal vaccines as well are produced using roller bottles. Tide Motion® Bioreactors mimic the rolling motion of Roller bottles which is an alternate emerging and submerging of cells for oxygenation and nutrient exchange.
By virtue of this, we offer a viable alternative to Roller bottle culture. This could be a less expensive and time –consuming as opposed to bioengineering adherent cell lines to suspension cells which may entail redoing of large-scale clinical trials and subsequent issues from a regulatory standpoint.

Tide Motion® Bioreactors
in Cell Culture Applications
The other very important application of Tide Motion® Bioreactors is in stem cell culture. Existing commercial and late stage allogeneic mesenchymal stem cell or mesenchymal stromal or medicinal signaling cells are often manufactured using either 2D multilayer planar culture ware with optional automated harvesting or 3D microcarriers which are occasionally dissolvable but often not.
Esco Aster provides Tide Motion® method as an alternative to linearly aid in scaling up MSCs for allogeneic cell therapy. The Tide Motion platform can also be used to produce allogeneic cell sources for gene- edited cell therapy as such CAR-T, CAR-NK as well as other immune cells that are derived from iPSCs or hESCs. Thus, other adherent packed bed bioreactors, hyperflasks, hyperstacks can easily be replaced by the Tide Motion® platform to provide viral vectors, for gene therapy and oncolytic viruses or cancer vaccines which often utilize HEK 293T and other adherent cell lines as expression platforms.

Tide Motion® Bioreactors as the best
alternative to other bioreactors in the market
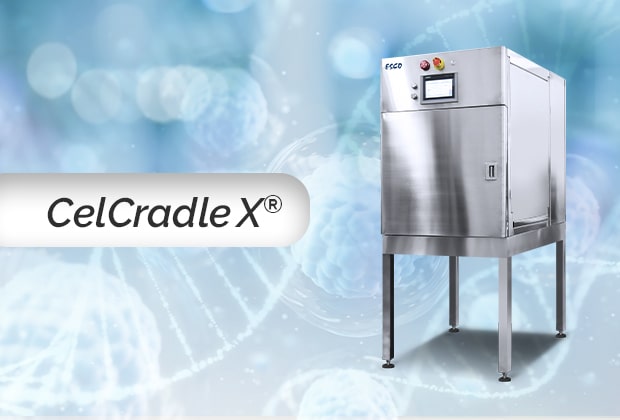
Our CelCradle™ X provides an alternative to other packed-bed bioreactors used for adherent autologous cell therapy ranging from Hollow Fiber bioreactors to automated multilayer culture bioreactors.
Analytical Chemistry Services
In addition to bioprocessing solutions, our analytical services utilizing analytical high performance liquid chromatography (HPLC), sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS-PAGE) and other analytical tools ensure that the Drug Substance and Drug Product are compatible and thus ensures equivalency. Other in vitro and in vivo techniques such as binding affinity utilizing Attana and BioCore are also utilized.

Bridging Studies and
Drug Product Manufacturing Services
Our clinical trial and commercial sites are able to produce Drug Products for bridging studies in a variety of container formats with our own quality control and lot release including sterility and stability testing.
