Construction of the Plasmids with Gene of Interest (GOI)
Simplify your plasmid design work with us. Esco Aster offers well-established platform for plasmid design and construction. Our capabilities include:
- expression vector design, for CAR constructs: scFv linker and spacer, transmembrane domain, choice of co-stimulatory domain(s)
- construct optimization for best packaging and expression
- platform switch guidance (for example from transposon or electroporation system to LV) with option to clone customer’s GOI into our proprietary backbone
- option to use our proprietary set of helper plasmids
Stable Cell Line Generation
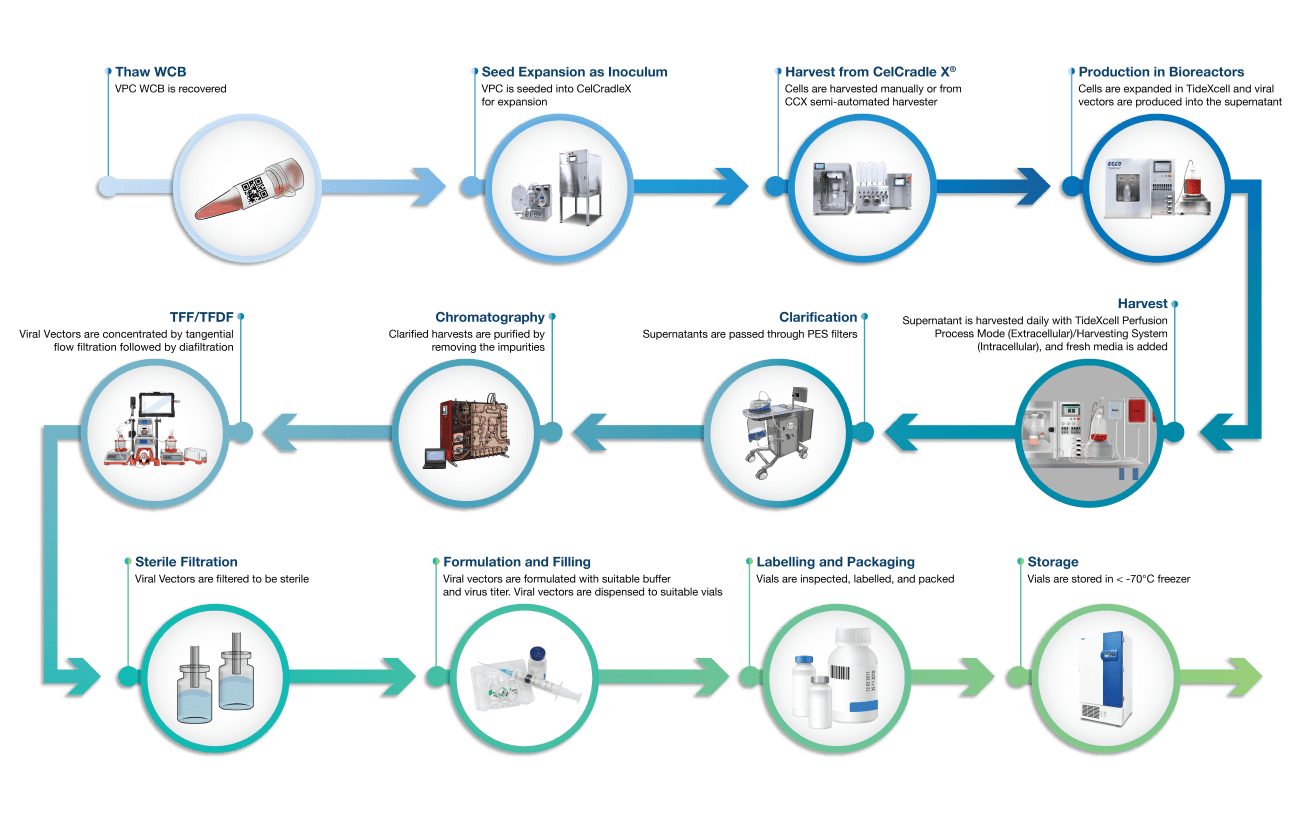
A high-quality and performance production cell line is the critical first step in any biopharmaceutical process. At Esco Aster, we have the ability to generate stable cell lines customized for your needs by implementing state-of-the-art single-cell cloning and high-resolution imager with full documentation to ensure monoclonality and to shorten the time of single cell cloning. We perform the process development with our proprietary bioreactors from lab-scale to manufacturing scale.


 650-min.jpg)

