Research-grade AAV
Production Services
Esco Aster’s team of scientific experts will work with you to guarantee that you receive AAV service that is customized to your research at every stage, meeting your needs quickly and efficiently. The features of this service comprise of:
- Safer, helper-independent AAV vector with helper-free expression system
- Thoroughly clarified and purified AAV vector with Esco Aster’s proprietary extraction method
- Several options of purification method to match your application/s
- Scale-up development for future commercial manufacturing

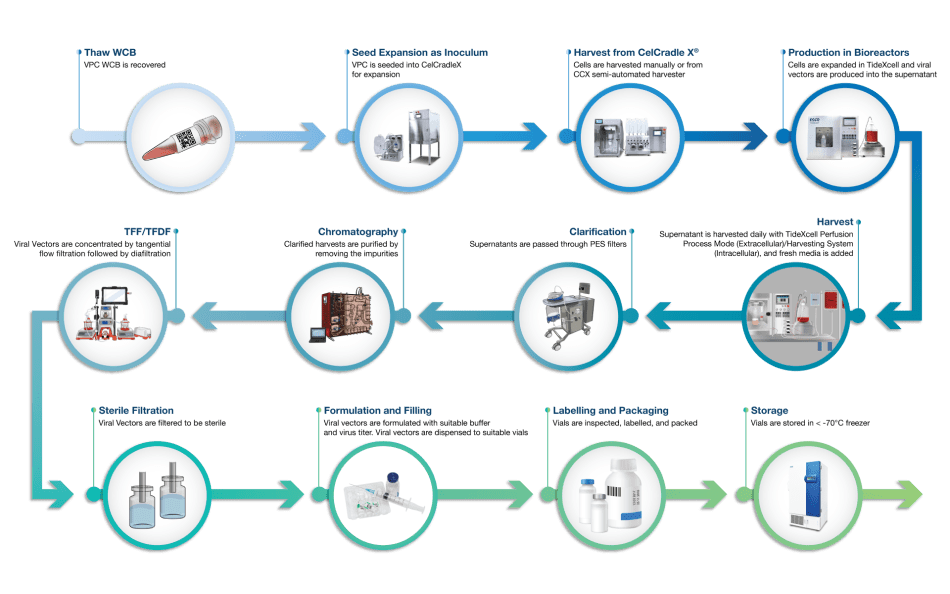
AAV’s Upstream Bioprocessing Platform
At Esco Aster, we consider many variables for adeno-associated virus (AAV) upstream bioprocess development. Within each step there are many ways to optimize and refine your process--a process that works, and a process that holds the promise of harvesting great results!
| Feature | Transfection | Sf9/Baculovirus | Producer Cell Line | HSV Co-Infection |
| Specialized Raw Materials |
|
|
|
|
| Scalability Concerns |
|
|
|
|
| Impurity Profile Concerns |
|
|
|
|
| Cell Culture System |
|
|
|
|
| Time Consideration |
|
|
|
|
Also available via client's technology transfer protocol
Clinical-grade AAV
Production Services
AAV vectors can transduce into proliferating cells and impart long-term expression. Furthermore, AAV vectors have very low immunogenicity. For these reasons, interest in the use of AAV vectors in gene therapy is rapidly growing. As each serotype shows different characteristics and tissue specificity, AAV vectors are expected to transduce target genes efficiently into heart, liver, muscle, retina, and central nervous system tissues.
- Project Initiation: We will be requiring plasmid DNA (transfer plasmid, packaging plasmids) and a producer cell line
- Deliverables: We will deliver AAV vector, manufacturing report, and certificate of analysis
Moreover, we use a validated assay for measuring physical titer (genome copies) for all AAV packaging services. Titering is performed using qPCR with primers targeting the ITRs present in the viral genome and quantified by comparison to a standard curve of a plasmid sample of known concentration.

Commercial-grade AAV
Production Services
Esco Aster’s state-of-the-art cGMP facility provides the services of viral vector manufacturing for clinical materials and commercial products. The maximum manufacturing capacity using the Tide Motion technology can reach as high as 200 L packed bed volume of BioNOC™ II macrocarriers, equivalent to 1,000 L daily harvest volume. It will fully meet the production capacity of the commercial manufacturing.
Continued Process Verification (CPV) is in place, enabling the detection of variation in the manufacturing process and providing opportunities to proactively control variation and assuring that during routine production the process remains in a state of control.

Viral Vector Analytics
We offer a range of assays to characterize your vectors. We analyze the identity, potency, stability and safety of your vector using analytical tools such as flow cytometry, quantitative PCR, digital PCR, ELISA and particle analysis.