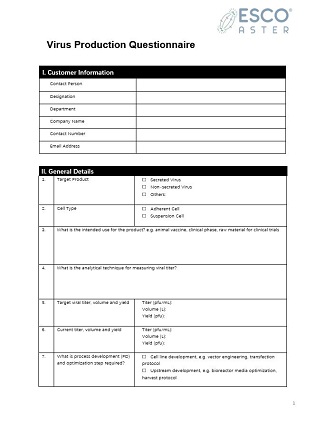
Aster Xpress™
Esco’s mobile diagnostic test laboratory and research solutions are designed for virus infected areas where specialized laboratory mechanical contractors are not present.
These turnkey services make the diagnostic unit easy to deploy in situations where the medical provider desires to cover large areas and in situations where mobility of health services is desirable (through shifting demand).



-min.jpg)